Chemical EquilibriumHard
Question
The equilibrium constant for the reaction N2(g) + O2(g) ⇋ 2NO(g) at temperature T is 4 × 10-4. The value of Kc for the reaction NO(g) ⇋ × 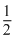 N2(g) +
N2(g) + 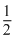 O2(g) at the same temperature is
O2(g) at the same temperature is
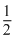 N2(g) +
N2(g) + 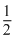 O2(g) at the same temperature is
O2(g) at the same temperature isOptions
A.2.5 × 102
B.0.02
C.4 × 10-4
D.50
Solution
As per property of equilibria reverse the equation & divide it by 2
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
For the reaction CaCO3(s) ⇋ CaO(s) + CO2(g), the pressure of CO2(g) depends on :...In the preceeding problem, if [A+] and [AB2-] are y and x respectively, under equilibrium produced by adding the substan...For the reversible reaction, N2(g) + 3H2(g) ⇋ 2NH3(g) + Heat The equilibrium shifts in forward direction...A sample of pure NO2 gas heated to 1000 K decomposes : 2NO2(g) ⇋ 2NO(g) + O2(g). The equilibrium constant KP is 10...Calculate equilibrium constant (approx) at 25oC for the cell reaction Fe(s) + CuSO4 ⇋ FeSO4 + Cu(s)Given : EoFe/Fe...