Chemical EquilibriumHard
Question
In the preceeding problem, if [A+] and [AB2-] are y and x respectively, under equilibrium produced by adding the substance AB to the solvents, then K1/K2 is equal to
Options
A.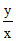 (y - x)2
(y - x)2
B.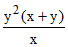
C.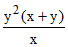
D.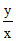 (x - y)
(x - y)
Solution
AB ⇋ A++ B- AB + B- ⇋ AB2-
a-x-y y (y-x) (a-x-y) y-x x
K1 =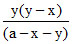 K2 =
K2 = 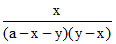
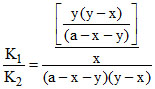 ⇒
⇒ 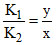 (y - x)2
(y - x)2
a-x-y y (y-x) (a-x-y) y-x x
K1 =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
XY2 dissociates XY2(g) ⇋ XY(g) + Y(g). When the initial pressure of XY2 is 600 mm Hg, the total equilibrium pressu...Two gases A and B, one being the dimer of the other, are at equilibrium. Decreasing of pressure at constant temperature ...At chemical equilibrium, the...Which of the following lines correctly show the temperature dependence of equilibrium constant, K, for an exothermic rea...1 mole each of H2(g) and I2(g) are introduced in a 1L evacuated vessel at 523K and equilibrium H2(g) + I2(g) ⇋ 2HI...