Chemical EquilibriumHard
Question
A sample of pure NO2 gas heated to 1000 K decomposes : 2NO2(g) ⇋ 2NO(g) + O2(g). The equilibrium constant KP is 100 atm. Analysis shows that the partial pressure of O2 is 0.25 atm. at equilibrium. The partial pressure of NO2 at equilibrium is:
Options
A.0.03
B.0.25
C.0.025
D.0.04
Solution
2NO2 ⇋ 2NO(g) + O2(g)
Kp =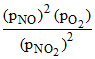
given PO2 = 0.25 ; PNO2 = 0.25
100 =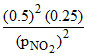
(PNO2)2 =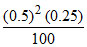
PNO2 = 0.025
Kp =
given PO2 = 0.25 ; PNO2 = 0.25
100 =
(PNO2)2 =
PNO2 = 0.025
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
A reaction at 300 K with ΔGo = −1743 J consists of 3 moles of A(g), 6 moles of B(g) and 3 moles of C(g). If A, B and C a...Given that the equilibrium constant for the reaction 2SO2(g) + O2(g) ⇋ 2SO3(g) has a value of 278 at a particular ...The equilibrium mixture for the reaction 2H2S(g)$\rightleftharpoons$ 2H2(g) + S2(g) has 1 mole of H2S, 0.20 mole of H2 a...In a system A(s) $\rightleftharpoons$2B(g) + 3C(g), if the concentration of C at equilibrium is increased by a factor of...Select incorrect reaction which is related to prepration of Cl2(g) -...