Chemical EquilibriumHard
Question
Calculate equilibrium constant (approx) at 25oC for the cell reaction
Fe(s) + CuSO4 ⇋ FeSO4 + Cu(s)
Given : EoFe/Fe+2 = + 0.453 volt ;
EoCu/Cu+2 = - 0.3435 volt
Fe(s) + CuSO4 ⇋ FeSO4 + Cu(s)
Given : EoFe/Fe+2 = + 0.453 volt ;
EoCu/Cu+2 = - 0.3435 volt
Options
A.1.0 × 1025
B.1.0 × 1020
C.1.0 × 1027
D.1.0 × 1022
Solution
Eocell = (EoRP)cathode - (EoRP)anode
Eocell = 0.3435 - (-0.453)
Eocell = 0.3435 + 0.453
= 0.7965 volt
Eocell =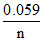 log Keq
log Keq
log Keq =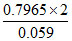 = 27 ⇒ Keq = 1027
= 27 ⇒ Keq = 1027
Eocell = 0.3435 - (-0.453)
Eocell = 0.3435 + 0.453
= 0.7965 volt
Eocell =
log Keq =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
1 mole each of H2(g) and I2(g) are introduced in a 1L evacuated vessel at 523K and equilibrium H2(g) + I2(g) ⇋ 2HI...The equilibrium constant for the reaction CO(g) + H2O(g) $\rightleftharpoons$CO2(g) + H2(g) is 3.0 at 500 K. In a 2.0 L ...An industrial fuel, ′water gas′ , which consists of a mixture of H2 and CO can be made by passing steam over...The correct relationship between free energy change in a reaction and the corresponding equilibrium constant K is...The system PCl5(g) ⇋ PCl3(g) + Cl2(g) attains equilibrium. If the equilibrium cocentrateion of PCl3(g) is doubled,...