Ionic EquilibriumHard
Question
The conjugate base of H2PO4- is
Options
A.PO43-
B.HPO42-
C.H3PO4
D.P2O5
Solution
∵ 6.20 × 10+20 molecules of urea is present in 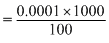 = 0.0 1M
= 0.0 1M
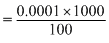 = 0.0 1M
= 0.0 1MCreate a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Choose the correct statement:...Dissociation constants of acids HA and HB are 2.0 × 10−4 and 5 × 10−5, respectively. The [H+] in the resulting solution ...The pKa of acetyl salicylic acid (aspirin) is 3.5. The pH of gastric juice in human stomach is about 2-3 and the pH in t...When 0.1 mole solid NaOH is added in 1lt of 0.1 M NH3(aq) then which statement is wrong? (Kb = 2 × 10-5, log 2 = 0....Sodium nitroprusside when added to an alkaline solution of sulphide ions produce a :-...