Ionic EquilibriumHard
Question
When 0.1 mole solid NaOH is added in 1lt of 0.1 M NH3(aq) then which statement is wrong?
(Kb = 2 × 10-5, log 2 = 0.3)
(Kb = 2 × 10-5, log 2 = 0.3)
Options
A.degree of dissociation of NH3 approaches to zero.
B.change in pH by adding NaOH would be 1.85
C.In solution, [Na+] = 0.1 M, [NH3] = 0.1 M, [OH-] = 0.2 M.
D.on addition of OH-, Kb of NH3 does not changes.
Solution
Initial pH = 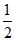 (pKb - log C) =
(pKb - log C) = 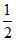 (5 - log 2 - log 0.1) = 2.85
(5 - log 2 - log 0.1) = 2.85
After adding NaOH, pOH of solution = 1
Change in pOH = 1.85
After adding NaOH, pOH of solution = 1
Change in pOH = 1.85
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Ka values for HA, HB and HD are 10-5, 10-7 and 10-9 respectively. Which of the following will be correct for decimolar a...To 100 ml of a solution, which contains 8.32 × 10–3 g lead ions, 10−4 moles of H2SO4 is added. How much lead remains in ...A 0.1 M solution of [Cu(NH3)4]+ is stirred with an excess of potassium cyanide sufficient to convert all the ammonium co...What is the equilibrium constant of the following reaction? Fe(OH)3(s) + 3H3O+ $\rightleftharpoons$ Fe3+ + 6H2O?Ksp of ...The $pK_a$ of a weak acid (HA) is 4.5. The pOH of an aqueous buffered solution of HA in which 50% of the acid is ionized...