Ionic EquilibriumHard
Question
Choose the correct statement:
Options
A.pH of acidic buffer solution decreases if more salt is added
B.pH of acidic solution increases if more salt is added.
C.pH of basic buffer decreases if more salt is added.
D.pH of basic buffer increases if more salt is added.
Solution
Acidic buffer: pH = pKa + log 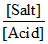 If more salt ius added, pH increases.
If more salt ius added, pH increases.
Basic buffer: pOH = pKb + log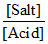 If more salt is added, pOH increases or pH decreases.
If more salt is added, pOH increases or pH decreases.
Basic buffer: pOH = pKb + log
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
A certain weak acid has a dissociation constant of 1.0 × 10-4. The equilibrium constant for its reaction with a str...Which is the correct statement for the given acids?...Calculate the formation constant for the reaction of a tri-positive metal ion with thiocyanate ions to form the monocomp...Which of the following when added to 1.0 L of 0.5 M HCl would result in maximum increase in pH :-...A tetrapeptide on partial hydrolysis produces a dipeptide (Gly-Ala or Ala-Gly) with two other amino acids. On complete h...