NEETSolid StateHard
Question
Following three planes( P1, P2, P3) in an FCC unit cell are shown:
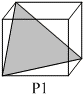
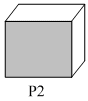
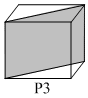
Consider the following statements and choose the correct option that follow:
(i) P1 contains no voids of three dimensions.
(ii) P2 contains only Octahedral voids.
(iii) P3 contains both Octahedral and Tetrahedral voids.



Consider the following statements and choose the correct option that follow:
(i) P1 contains no voids of three dimensions.
(ii) P2 contains only Octahedral voids.
(iii) P3 contains both Octahedral and Tetrahedral voids.
Options
A.All are true
B.Only (i) & (ii) are true
C.(i) & (iii) are true
D.Only (iii) is true.
Solution
Refer theory octahedral & tetrahedral voids about positions of
Create a free account to view solution
View Solution FreeMore Solid State Questions
A crystal of NaCl, which has sodium ion and chloride ion missing from the lattice point, is said to exhibit...Sodium metal crystallizes in a body centred cubic lattice with a unit cell edge of 4.29(Ao). The radius of sodium atom i...TiO2 is a well-known example of...Which of the following are the correct axial distance and axial angles for rhombohedral system?......