Solid StateHard
Question
How many unit cells are present in a cubeshaped ideal crystal of NaCl of mass 1.00 g?
[Atomic masses: Na = 23, Cl = 35.5]
[Atomic masses: Na = 23, Cl = 35.5]
Options
A.5.14 × 1021 unit cells
B.1.28 × 1021 unit cells
C.1.71 × 1021 unit cells
D.2.57 × 1021 unit cells
Solution
Number of formulas in cube shaped crystals = 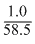 × 6.02 × 1023 since in NaCl type of structure 4 formula units form a cell
× 6.02 × 1023 since in NaCl type of structure 4 formula units form a cell
∴ unit cells =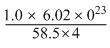 = 2.57 × 1021 unit cells.
= 2.57 × 1021 unit cells.
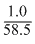 × 6.02 × 1023 since in NaCl type of structure 4 formula units form a cell
× 6.02 × 1023 since in NaCl type of structure 4 formula units form a cell∴ unit cells =
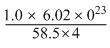 = 2.57 × 1021 unit cells.
= 2.57 × 1021 unit cells.Create a free account to view solution
View Solution FreeMore Solid State Questions
Platinum crystallises in a face centered cube crystal with a unit cell length of 3.9231 A0. The density and atomic radiu...Which of the following may have Frenkel defect?...In a face centerd lattice of X and Y, X atoms are present at the corners while Y atoms are at face centers. Then the for...Calcium titanate crystallizes in a cubic unit cell with titanium atoms at the corner of the cell, oxygen atoms in the mi...Which of the following oxides behave as a conductor or an insulator depending on temperature?...