Chemical Kinetics and Nuclear ChemistryHard
Question
The first order rate constant for a certain reaction increase from 1.667 × 10-6 s-1 at 727oC to 1.66 × 10-4s-1 at 1571oC. The rate constant at 1150oC assuming constancy of activation energy over the given temprature range is
[Given : log 19.9 = 1.299][2009]
[Given : log 19.9 = 1.299][2009]
Options
A.3.911 × 10-5s-1
B.1.139 × 10-5s-1
C.3.318 × 10-5s-1
D.1.193 × 10-5s-1
Solution
log 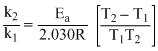
or 2.030 log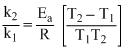
2.030 log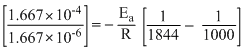
2.030 × 2 =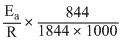 .......(1)
.......(1)
∴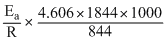
2.030log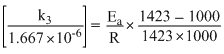
=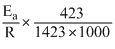 ......(2)
......(2)
Dividing equation (2)by equation (1)
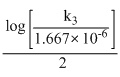
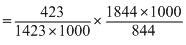
∴ log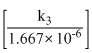
2 ×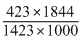 = 1.299
= 1.299
On taking Antilog., k3 = 19.9
∴ k3 = 19.9 × 1.667 × 10-6 = 3.318 × 10-5s-1
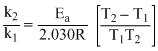
or 2.030 log
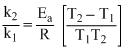
2.030 log
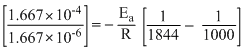
2.030 × 2 =
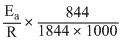 .......(1)
.......(1)∴
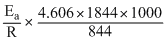
2.030log
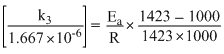
=
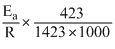 ......(2)
......(2)Dividing equation (2)by equation (1)
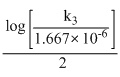
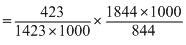
∴ log
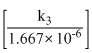
2 ×
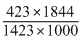 = 1.299
= 1.299On taking Antilog., k3 = 19.9
∴ k3 = 19.9 × 1.667 × 10-6 = 3.318 × 10-5s-1
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
In compounds of type ECl3 where E = B, P. As or Bi, the angles Cl − E − Cl for different R in order-...Following are the values of Ea and DH for three reactions carried out at the same temperature : I : Ea = 20 kJ mol-1, &#...A reaction was found to be second order with respect to the concentration of carbon monoxide. If the concentration of ca...What is ᐃH for the reaction A + B → C, where the mechanism involves several kinetic steps....Number of Faraday required to liberate 8 g. of H2 is :-...