JEE Advanced | 2014Chemical Kinetics and Nuclear ChemistryHard
Question
Isomers of hexane, based on their branching, can be divided into three distinct classes as shown in the figure.
The correct order of their boiling point is
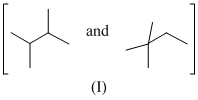
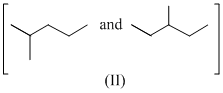
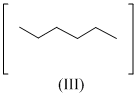
The correct order of their boiling point is

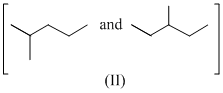

Options
A.I > II > III
B.III > II > I
C.II > III > I
D.III > I > II
Solution
As branching increases boiling point decreases, so order of boiling point is III > II > I.
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Consider the reaction : Cl2(aq) + H2S(aq) → S(s) + 2H+(aq) + 2Cl- (aq) The rate equation for this reaction is rate...For a process A+ B Products, the rate is first order with respect to A and First order with respect to B. When 1.0 mol e...In a reaction, A + B → Product, rate is doubled when the concentration of B is doubled, and rateincreases by a fac...Which of the following is a lyophobiccolloidal solution?...For two reactions $A\overset{\quad K_{1}\quad}{\rightarrow}P\text{ and }B\overset{\quad K_{2}\quad}{\rightarrow}Q$ of fi...