ElectrochemistryHard
Question
Given the data at 25oC,
Ag + I- → Agl + e- ; Eo = 0.152 V
Ag → Ag+ + e- : Eo = - 0.800 V
What is the value of log Ksp for AgI ?
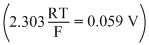
Ag + I- → Agl + e- ; Eo = 0.152 V
Ag → Ag+ + e- : Eo = - 0.800 V
What is the value of log Ksp for AgI ?
Options
A.- 8.12
B.+8.612
C.- 37.83
D.- 16.13
Solution
AgI(s) + e- ⇋ Ag(s) + I- ; Eo = - 0.152
Ag(s) → Ag+ + I- ; Eo = - 0.8
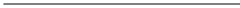
AgI(s) → Ag+ + I- ; Eo = - 0.952
Eocell =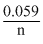 log K
log K
- 0.952 =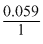 log Ksp
log Ksp
log Ksp = -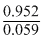 = - 16.135
= - 16.135
Ag(s) → Ag+ + I- ; Eo = - 0.8
AgI(s) → Ag+ + I- ; Eo = - 0.952
Eocell =
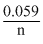 log K
log K- 0.952 =
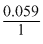 log Ksp
log Ksplog Ksp = -
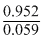 = - 16.135
= - 16.135Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The molar conductance of a 0.01 M solution of acetic acid was found to be 16.30 Ω−1 cm−1 mol−1 at 25°C. The ionic conduc...If 0.224 L of H2 gas is formed at the cathode, the volume of O2 gas formed at the anode under identical conditions, is...1/2 H2(g) + AgCl(s) = H+ (aq) + Cl- (aq) + Ag (s) occurs in the galvanic cell :...Assuming that hydrogen behaves as an ideal gas, what is the EMF of the cell at 25°C if P1 = 600 mm and P2 = 420 mm: Pt |...By how much will the potential of half cell Cu2+/ Cu change if the solution is diluted to 100 times at 298 K...