ElectrochemistryHard
Question
If 0.224 L of H2 gas is formed at the cathode, the volume of O2 gas formed at the anode under identical conditions, is
Options
A.0.224 L
B.0.448 L
C.0.112 L
D.1.12 L
Solution
equivalence of H2 = equivalence of O2
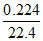 × 2 =
× 2 = 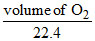 × 4
× 4
0.112 litre = volume of O2.
0.112 litre = volume of O2.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Select the correct statement.(i) Delocalisation of σ-electron is hyperconjugation.(ii) Delocalisation of π-el...Which of the following is/are function(s) of salt - bridge ?...Which of the following molecules can act as an oxidizing as well as a reducing agent ?...Peroxodisulphate salts (e.g., Na2S2O8) are storng oxidizing agents used as bleaching agents for fats, oils, etc.Given: O...The charge required for the reduction of 1 mol of MnO4- to MnO2 is :...