SolutionHardBloom L3
Question
A 5.25 % solution of a substance is isotonic with a 1.5% solution of urea (molar mass = 60 g mol-1) in the same solvent. If the densities of both the solutions are assumed to be equal to 1.0 g cm-3, molar mass of the substance will be
Options
A.90.0 g mol-1
B.115.0 g mol-1
C.105.0 g mol-1
D.210.0 g mol-1
Solution
Solutions with the same osmotic pressure are isotonic
Let the molar mass of the substance be M
π1 = C1RT = C2RT = π2
So, C1 = C2
As density of the solutions are same
So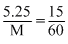
M =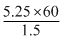 = 210
= 210
Hence (4) is correct
Let the molar mass of the substance be M
π1 = C1RT = C2RT = π2
So, C1 = C2
As density of the solutions are same
So
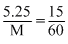
M =
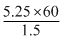 = 210
= 210Hence (4) is correct
Create a free account to view solution
View Solution FreeMore Solution Questions
When H2S gas is passed through the HCl containing aqueous solution of CuCl2, HgCl2, BiCl3, and CoCl2, Which does not pre...In the following aqueous solutions(A) 1 m sucrose (B) 1 m potassium ferricyanide andC) 1 m potassium sulphate maximum va...Depression of freezing point of 0.01 molal aq. CH3COOH solution is 0.02046o. 1 molal urea solution freezes at - 1.86°C. ...Observe the P-T phase diagram for a given substance A. Then melting point of A(s), boiling point of A(l), critical point...The vapor pressures of benzene, toluene and a xylene are 75 Torr, 22 Torr and 10 Torr at 20oC. Which of the following is...