SolutionHard
Question
Depression of freezing point of 0.01 molal aq. CH3COOH solution is 0.02046o. 1 molal urea solution freezes at - 1.86°C. Assuming molality equal to molarity, pH of CH3COOH solution is :
Options
A.2
B.3
C.3.2
D.4.2
Solution
For urea, ᐃTf = kf × m or kf = 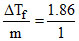 = 1.86
= 1.86
Now for CH3COOH
DTf = i kf m
so i =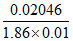 = 1.1
= 1.1
Now i = 1 + α
so α = 1.1 - 1 = 0.1
Now CH3COOH ⇋ CH3COO- + H+
C 0 0
C – Cα Cα Cα
[H+] = Cα = 0.01 × 0.1 = 0.001
so pH = 3.
Now for CH3COOH
DTf = i kf m
so i =
Now i = 1 + α
so α = 1.1 - 1 = 0.1
Now CH3COOH ⇋ CH3COO- + H+
C 0 0
C – Cα Cα Cα
[H+] = Cα = 0.01 × 0.1 = 0.001
so pH = 3.
Create a free account to view solution
View Solution FreeMore Solution Questions
The phase diagrams for the pure solvent (solid lines) and the solution (non-volatile solute, dashed line) are recorded b...Which one of the statements given below concerning properties of solutions, describes a colligative effect ?...If α is the degree of dissociation of Na2SO4, the vant Hoff′s factor (i) used for calculating the molecular m...On mixing 3g of non-volatile solute in 200 mL of water, its Boiling point (100oC) be comes 100.52oC. If Kb for water is ...Glucose in blood can be quantitatively determined with :-...