SolutionHard
Question
The vapor pressures of benzene, toluene and a xylene are 75 Torr, 22 Torr and 10 Torr at 20oC. Which of the following is not a possible value of the vapor pressure of an equimolar binary/ternary solution of these at 20oC ? Assume all form ideal solution with each other.
Options
A.48 1/2
B.16
C.35 2/3
D.53 1/2
Solution
Possible vapor pressures are
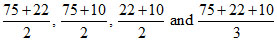
= 48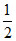 , 42
, 42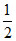 , 16, 35
, 16, 35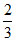 .
.
= 48
Create a free account to view solution
View Solution FreeMore Solution Questions
A current of 2.6 ampere was passed through CuSO4 solution for 380 sec. The amount of Cu deposited is (at. wt. of Cu = 63...The boiling point of an azeotropic mixture of water and ethyl alcohol is less than that of theoretical value of water an...18 g of glucose (C6H12O6) is added to 178.2 g of water. The vapour pressure of water for this aqueous solution at 100oC ...Consider following cases -I : 2M CH3COOH solution in benzene at 27oC where there is dimer formation to the extent of 100...The dissolving process is exothermic when :...