Chemical Kinetics and Nuclear ChemistryHard
Question
The energies of activation for forward and reverse reactions for A2 + B2 ⇋ 2AB are 180 kJ mol-1 2AB are 180 kJ mol-1 respectively. The presence of catalyst lowers the activation energy of both (forward and reverse) reactions by 100 kJ mol-1. The enthalpy change of the reaction (A2 + B2 → 2AB) in the presence of catalyst will be (in kJ mol-1)
Options
A.300
B.120
C.280
D.20
Solution
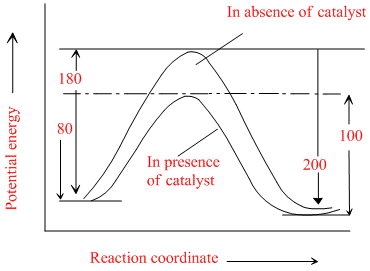
So, ᐃHReaction = Ef - Eb
= 80 - 100 = - 20
Hence, (4) is correct.
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Isomers of hexane, based on their branching, can be divided into three distinct classes as shown in the figure. The corr...A catalyst lowers the activation energy of a reaction from 20 kJ mol-1 to 10 kJ mol-1. The temperature at which the unca...A sample of 53I131, as I- ion, was administered to a patient in a carrier consisting 1.0 mg of stable I- ion. After 4.0 ...Match the graphical study with the order of the reactions : I : Rate II : Half life III : (a - x)-1...Purple of cassius is colloidal solution of :...