Chemical Kinetics and Nuclear ChemistryHard
Question
A sample of 53I131, as I- ion, was administered to a patient in a carrier consisting 1.0 mg of stable I- ion. After 4.0 days. 60% of the initial radioactivity was detected in the thyroid gland of the patient. What mass of the stable I- ion had migrated to the thyroid gland ? Given: t1/2 of I131 = 8 days
Options
A.0.65 mg
B.0.75 mg
C.0.85 mg
D.0.95 mg
Solution
k = 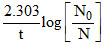
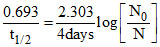
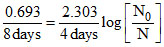
∴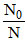 ≈ 0.7 ⇒ 70% of the initial activity is present.
≈ 0.7 ⇒ 70% of the initial activity is present.
Given that 60% activity is migrated to thyroid gland. weight of I- ion migrated to thyroid gland is:
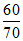 × 100 ≈ 05.7%
× 100 ≈ 05.7%
i.e., 1.0 mg ×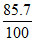 = 0.857 mg
= 0.857 mg
∴
Given that 60% activity is migrated to thyroid gland. weight of I- ion migrated to thyroid gland is:
i.e., 1.0 mg ×
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
For a first order reaction A → P, the temperature (T) dependent rate constant (k) was found to follow the equation...Which of the following statements are true for chemisorption...For the following reaction(CH3)3CCl + H2O → (CH3)3 COH + HCl, = k[CH3)3CCl], hence, rate determining step can be:...Units of rate constant of first and zero order reactions in terms of molarity M unit are respectively...For the chemical reaction A + 2B → C, rate of the reaction is increased by 2.82 times when the concentration of A ...