Atomic StructureHard
Question
If the binding energy of the electron in a hydrogen atom is 13.6 eV, the energy required to remove the electron from the first excited state of Li++ is
Options
A.30.6 eV
B.13.6 eV
C.3.4 eV
D.122.4 eV.
Solution
The energy of the first excited state of Li++ is
E2 = -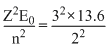 = - 30.6 eV.
= - 30.6 eV.
E2 = -
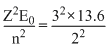 = - 30.6 eV.
= - 30.6 eV.Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The equation ; 411H → 24He2 + 2e- + 26MeV represents...Consider an atom made up of a proton and a hypothetical particle that has double the mass of an electron but the same ch...In hydrogen atom spectrum, ( $R \rightarrow$ Rydberg's constant)A. the maximum wavelength of the radiation of Lyman seri...The flux of α - particle 2o is 1 × 106. The flux of α - particle at angle 60o is...A electron in the hydrogen atom jumped from excited state n to the ground state. The wavelength so emitted illuminates a...