Atomic StructureHard
Question
The flux of α - particle 2o is 1 × 106. The flux of α - particle at angle 60o is
Options
A.1.5
B.2.5
C.0.5
D.5.5
Solution
N(θ) ∝ 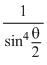
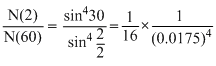
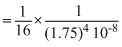
N(60) = 16 × (1.75)4 × 10-8 × N(2)
= 16 × (1.75)4 × 10-8 × 106
= 16 × (1.75)4 × 10-2 = 1.5
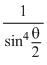
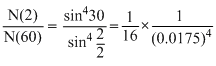
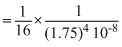
N(60) = 16 × (1.75)4 × 10-8 × N(2)
= 16 × (1.75)4 × 10-8 × 106
= 16 × (1.75)4 × 10-2 = 1.5
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The difference between the longest wavelength line of the Balmer series and shortest wavelength line of the Lyman series...If the velocity of light were (2/3)rd of the present value, then the energy released in atomic explosion will be reduced...Shown in the figure are two point charges +Q and - Q inside the cavity of a spherical shell. The charges arekept near th...Half of a substance is 20 minutes, then the time between 33% decay and 67% decay will be...Two hydrogen atoms are in excited state with electrons residing in n = 2. First one is moving towards left and emits a p...