Atomic StructureHard
Question
A electron in the hydrogen atom jumped from excited state n to the ground state. The wavelength so emitted illuminates a photosensitive material having work function 2.75 eV. If the stopping potential of the photoelectron is 10 V, then the value of n is
Options
A.2
B.3
C.4
D.5
Solution
Here, stopping potential, V0 = 10V
Work function, W = 2.75 eV
According to Einstein′s photoelectric equation
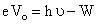
or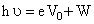
= 10 eV + 2.75 eV = 12.75 eV ...... (i)
When an electron in the hydrogen atom makes a transition from excited state n to the ground state (n = 1), then he frequency (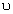 ) of the emitted photon is given by
) of the emitted photon is given by
h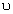 = En - E1
= En - E1
h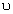 = -
= - 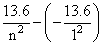
[∵ For hydrogen atom, En =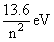 ]
]
According to the given problem
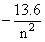 + 13.6 = 12.75 [Using (i)]
+ 13.6 = 12.75 [Using (i)]
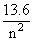 = 0.85 ⇒ n2 =
= 0.85 ⇒ n2 = 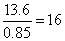
or n = 4
Work function, W = 2.75 eV
According to Einstein′s photoelectric equation
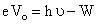
or
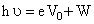
= 10 eV + 2.75 eV = 12.75 eV ...... (i)
When an electron in the hydrogen atom makes a transition from excited state n to the ground state (n = 1), then he frequency (
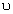 ) of the emitted photon is given by
) of the emitted photon is given by h
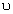 = En - E1
= En - E1h
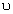 = -
= - 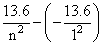
[∵ For hydrogen atom, En =
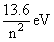 ]
]According to the given problem
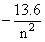 + 13.6 = 12.75 [Using (i)]
+ 13.6 = 12.75 [Using (i)]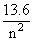 = 0.85 ⇒ n2 =
= 0.85 ⇒ n2 = 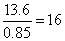
or n = 4
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
When photo energy 4.25 eV strike the surface of a metal A, the ejected photoelectrons have maximum kinetic energy TA exp...As per Bohr's model, the minimum energy (in eV) required to remove an electron from the ground state of doubly ionized L...A mixture of 2 moles of helium gas (atomic mass = 4 amu) and 1 mole of argon gas (atomic mass = 40 amu) is kept at 300 K...For an electron in the second orbit of hydrogen, the moment of momentum as per Bohr′s model is...If a source of power 4 kW produces 1020 photons/second, the radiation belong to a part of the spectrum called...