ElectrochemistryHard
Question
Consider the following cell reaction:
2Fe(s) + O2(g) + 4H+(aq) → 2Fe2+(aq) + 2H2O(l) Eo = 1.67V
At [Fe2+] = 10-3 M, P(O2) = 0.1 atm and pH = 3, the cell potential at 25oC is
2Fe(s) + O2(g) + 4H+(aq) → 2Fe2+(aq) + 2H2O(l) Eo = 1.67V
At [Fe2+] = 10-3 M, P(O2) = 0.1 atm and pH = 3, the cell potential at 25oC is
Options
A.1.47 V
B.1.77 V
C.1.87 V
D.1.57 V
Solution
2Fe (s) + O2(g) + 4H+ (aq) → 2Fe+2 (aq) + 2H2O(l)
N = 4 (no. of moles of electron involved)
From Nernst′s equation,
Ecell = Eocell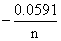 log Q
log Q
= 1.67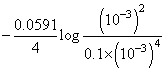 {∴ [H+] = 10-pH}
{∴ [H+] = 10-pH}
= 1.67 - 0.106 = 1.57 V
N = 4 (no. of moles of electron involved)
From Nernst′s equation,
Ecell = Eocell
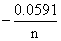 log Q
log Q= 1.67
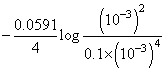 {∴ [H+] = 10-pH}
{∴ [H+] = 10-pH}= 1.67 - 0.106 = 1.57 V
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Saturated solution of KNO3 is used to make ′salt-bridge′ because :...In the electrolysis of CuCl2 soluton using Cu electrodes, the weight of Cu increased by 2gram at cathode. At the anode -...Cu+ + e- → Cu, Eo = x1 volt ;Cu2+ + 2e- Cu, Eo = x2 volt, then forCu2+ + e- Cu+, Eo (volt) will be -...Zn Amalgam is prepared by electrolysis of aqueous ZnCl2 using Hg cathode (9gm). How much current is to be passed through...On electrolysis, in which of the following, O2 would be liberated at the anode ?...