ElectrochemistryHard
Question
Zn Amalgam is prepared by electrolysis of aqueous ZnCl2 using Hg cathode (9gm). How much current is to be passed through ZnCl2 solution for 1000 seconds to prepare a Zn Amalgam with 25% Zn by wt. (Zn = 65.4)
Options
A.5.6 amp
B.7.2 amp
C.8.85 amp
D.11.2 amp
Solution
Let x gm of Zn deposit on 9 gm of Hg
% of Zn in Amalgam =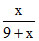 x 100 = 25
x 100 = 25
∴ x = 3gm
Eq. of Zn =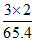
Current = x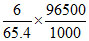 = 8.85 amp.
= 8.85 amp.
% of Zn in Amalgam =
∴ x = 3gm
Eq. of Zn =
Current = x
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Standard electrode potential of three metals X, Y, and Z are -1.2 V, + 0.5 V and -3.0 V respectively. The reducing power...Given EoAg+/Ag = 0.80V, EoMg2+/Mg = - 2.37V, EoCu2+/Cu = 0.34, EoHg2+/Hg = 0.79 V.Which of the following statements is/a...The cell Zn|Zn²⁺(1M)||Cu²⁺(1M)|Cu with $E^\circ_{\text{cell}} = 1.10$ V was allowed to be completely discharged at 298 K...For a spontaneous reaction the ᐃG, equilibrium constant (K) and Eocell will be respectively...At equimolar concentrations of Fe2+ and Fe3+, what must [Ag+] be so that the voltage of the galvanic cell made from the ...