Coordination CompoundHard
Question
The coordination number and the oxidation state of the element ′E′ in the complex [E(en)2(C2O4)]NO2
(where (en) is ethylene diamine) are, respectively,
(where (en) is ethylene diamine) are, respectively,
Options
A.6 and 2
B.4 and 2
C.4 and 3
D.6 and 3
Solution
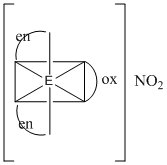
Coordination no. = 6 and Oxidation no. = 3
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Low spin complex is formed by :...Which of the following statements is correct for complex [Cr(NH3)(CN)4(NO)]2 - (given that n = 1)?...The tetrahedral [CoI4]2– and square planar [PdBr4]2– complex ions are respectively...Which of the following is a bidentate, unsymmaterical, anionic ligand :-...Which of the following complex ions is not expected to absorb visible light ?...