Coordination CompoundHard
Question
Which of the following complex ions is not expected to absorb visible light ?
Options
A.[Ni(CN)4]2-
B.[Cr(NH3)6]3+
C.[Fe(H2O)6]2+
D.[Ni(H2O)6]2+
Solution
For the absorption of visible light, presence of unpaired d-electrons is the necessity.
(a) In [Ni(CN)4]2-, Ni is present as Ni2+.
Ni2+ = [Ar] 3d8 4s0
∴ [Ni(CN)4]2- =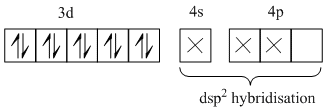
(Pairing occurs because CN is a strong field ligand).
Since, in [Ni(CN)4]2-, no unpaired electron is present in d-orbitals, it does not absorb visible light.
(b) In [Cr(NH3)6]3+, Cr is present as Cr3+ .
Cr3+ = [Ar]3d3 4s0 (Three unpaired electrons)
(c) In [Fe(H2O)6]2+, Fe is present as Fe2+.
Fe2+ = [Ar]3d6 4s0 (Four unpaired electrons)
(d) In [Ni (H2O)6]2+, Ni is present as Ni2+.
Ni2+ = [Ar] 3d8 4s0 (Two unpaired electrons)
The complexes given in option (b), (c), (d) have unpaired electrons, thus absorb visible light.
(a) In [Ni(CN)4]2-, Ni is present as Ni2+.
Ni2+ = [Ar] 3d8 4s0
∴ [Ni(CN)4]2- =

(Pairing occurs because CN is a strong field ligand).
Since, in [Ni(CN)4]2-, no unpaired electron is present in d-orbitals, it does not absorb visible light.
(b) In [Cr(NH3)6]3+, Cr is present as Cr3+ .
Cr3+ = [Ar]3d3 4s0 (Three unpaired electrons)
(c) In [Fe(H2O)6]2+, Fe is present as Fe2+.
Fe2+ = [Ar]3d6 4s0 (Four unpaired electrons)
(d) In [Ni (H2O)6]2+, Ni is present as Ni2+.
Ni2+ = [Ar] 3d8 4s0 (Two unpaired electrons)
The complexes given in option (b), (c), (d) have unpaired electrons, thus absorb visible light.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
The crystal field-splitting for Cr3+ ion in octahedral field changes for ligands I-, H2O, NH3, CN- and the increasing or...Coordination compounds have great importance in biological systems. In this context which of the following statements is...The complexes [Co (NH3 )6] [Cr (CN )6 ] and [Cr (NH3 )6 ] [Co (CN)3 ] are the examples of which type of isomerism ?...Red precipitate is obtained when ethanol solution of dimethylglyoxime is added to ammoniacal Ni(II).Which of the followi...The number of chloride ions which would be precipitated when the complex PtCl4.4NH3 is treated with silver nitrate is:(h...