Coordination CompoundHard
Question
Which of the following statements is correct for complex [Cr(NH3)(CN)4(NO)]2 - (given that n = 1)?
Options
A.It is d2sp3 hybridised
B.The chromium is in + I oxidation state
C.It is heteroleptic complex and its aqueous solution is coloured
D.All of these.
Solution
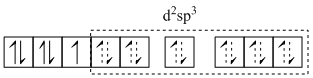
Central metal ion has one unpaired electron and thus d-d transition of electron is possible. This attributes to the colour of the solution. As different types of ligands are attached to central metal ion, the complex is called heteroleptic complex.
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Complexes [Co(NH3)5SO4]Br and [Co(NH3)5Br]SO4 can be distinguished by :...Which of the following exhibit geometrical isomerism (M stands for a metal, and a and b are achiral ligands)?...The complex used as an antixcancer agent is...Which ligand forms chelates :-...Which of the following complexes show geometrical as well as optical isomerism ?(1) [Cr(OX)3]3-(2) [Rh(en)2Cl2]+(3) [Co(...