ThermodynamicsHard
Question
In a fuel cell methanol is used as fuel and oxygen gas is used as an oxidizer. The reaction is -
CH3OH(l) + 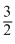 O2(g) → CO2(g) + 2H2O(l)
O2(g) → CO2(g) + 2H2O(l)
At 298K, standard Gibb’s energies of formation for CH3OH)(l), H2O(l) and CO2(g) are -166.2, -237.2 and -394.4 kJ mol-1 respectively. If standard enthalpy of combustion of methanol is -726kJ mol-1, Efficiency of the fuel cell will be
Options
A.80 %
B.87 %
C.90 %
D.97 %
Solution
CH3OH(l) + 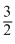 O2(g) + 2H2O(l) áƒH = - 726kJ mol-1
O2(g) + 2H2O(l) áƒH = - 726kJ mol-1
Also áƒGfo CH3OH(l) = -166.2 kJ mol-1
áƒGfoH2O(l) = -237.2 kJ mol-1
áƒGfoCO2(l) = -394.4 kJ mol-1
∵  áƒG = ΣáƒGfo products -ΣáƒGfo reactants.
= -394.4 -2 (237.2) + 166.2
= -702.6 kJ mol-1
now Efficiency of fuel cell =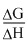 × 100
× 100
=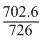 × 100
× 100
= 97%
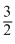 O2(g) + 2H2O(l) áƒH = - 726kJ mol-1
O2(g) + 2H2O(l) áƒH = - 726kJ mol-1 Also áƒGfo CH3OH(l) = -166.2 kJ mol-1
áƒGfoH2O(l) = -237.2 kJ mol-1
áƒGfoCO2(l) = -394.4 kJ mol-1
∵  áƒG = ΣáƒGfo products -ΣáƒGfo reactants.
= -394.4 -2 (237.2) + 166.2
= -702.6 kJ mol-1
now Efficiency of fuel cell =
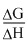 × 100
× 100=
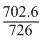 × 100
× 100= 97%
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
For a spontaneous process, entropy:...Standard entropies of X2, Y2 and XY3 are 60, 40 and 50 JK-1 mol-1 respectively. For the reaction y2 ⇋ XY3 ; ᐃ...At 300 K, the reactions which have the following values of thermodynamic parameters, occur spontaneously....1 mol of NH3 gas at 27oC is expanded under adiabatic condition to make volume 8 times (g = 1.33). Final temperature and ...one mole of an ideal gas is allowed to expand reversibly and adiabatically from a temperature of 27oC. if the work done ...