ThermodynamicsHard
Question
one mole of an ideal gas is allowed to expand reversibly and adiabatically from a temperature of 27oC. if the work done during the process is 3kJ, then final temperature of the gas is (Cv = 20J/K)
Options
A.100 K
B.150 K
C.195 K
D.255 K
Solution
Since the gas expands adiabatically (i.e., no change in enthalpy) so the heat is totallyconverted in to work
For the gas, Cv = 20J/K. Thes 20 J of heat is required for 1o change in temperature of the gas.Heat change involved during during the process i.e., work done = 3 kJ = 3000J.
Change in temperature =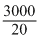 K = 150K
K = 150K
Initial temprature = 300 K
Since, the gas thus final temperrature decreases and thus final tempeerature is300 - 150 = 150
For the gas, Cv = 20J/K. Thes 20 J of heat is required for 1o change in temperature of the gas.Heat change involved during during the process i.e., work done = 3 kJ = 3000J.
Change in temperature =
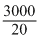 K = 150K
K = 150KInitial temprature = 300 K
Since, the gas thus final temperrature decreases and thus final tempeerature is300 - 150 = 150
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
One mole of a real gas is subjected to heating at constant volume from (P1, V1, T1) state to (P2, V1, T2) state. Then it...The standard Gibbs energy change at 300 K for the reaction 2A ⇋ B + C is 2494.2J. At a given time, the composition...An amount of 5 mole H2O(l) at 100o C and 1 atm is converted into H2O(g) at 100°C and 5 atm. ΔG for the process is...At very low temperature, the heat capacity of crystals is equal to C = aT3, where a is a constant. Find the entropy of a...Which of the following physical quantities are both independent from amount and path...