JEE AdvancedThermodynamicsHard
Question
Standard entropies of X2, Y2 and XY3 are 60, 40 and 50 JK-1 mol-1 respectively. For the reaction
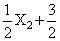 y2 ⇋ XY3 ; ᐃH = - 30kJ , to be at equilibrium, the temperature should be
y2 ⇋ XY3 ; ᐃH = - 30kJ , to be at equilibrium, the temperature should be
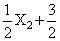 y2 ⇋ XY3 ; ᐃH = - 30kJ , to be at equilibrium, the temperature should be
y2 ⇋ XY3 ; ᐃH = - 30kJ , to be at equilibrium, the temperature should beOptions
A.750 K
B.1000 K
C.1250 K
D.500 K
Solution
For the reaction,
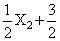 y2 ⇋ XY3 ; ᐃH = - 30kJ
y2 ⇋ XY3 ; ᐃH = - 30kJ
ᐃSo = So(XY3) -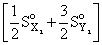
= 50 -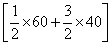
= 50 - [30 + 60]
= 50 - 90
= - 40 JK-1 mol-1
We know that,
ᐃGo = ᐃHo - T ᐃSo
At equilibrium,
ᐃGo = 0
ᐃH = TᐃSo
T =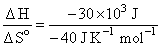
= 750 K
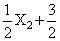 y2 ⇋ XY3 ; ᐃH = - 30kJ
y2 ⇋ XY3 ; ᐃH = - 30kJᐃSo = So(XY3) -
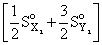
= 50 -
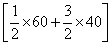
= 50 - [30 + 60]
= 50 - 90
= - 40 JK-1 mol-1
We know that,
ᐃGo = ᐃHo - T ᐃSo
At equilibrium,
ᐃGo = 0
ᐃH = TᐃSo
T =
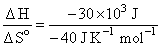
= 750 K
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
A piece of alloy weighing 4 kg and at a temperature of 800 K is placed in 4 kg of water at 300 K. If the specific heat c...Identify the correct statement regarding a spontaneous process...The magnitude of work done by one mole of a Van der Waals gas during its isothermal reversible expansion from volume V1 ...What is the free energy change, ΔG, when 1.0 mole of water at 100°C and 1 atm pressure is converted into steam at 100°C ...Which of the following pair does show the extensive properties?...