ThermodynamicsHard
Question
Two moles of helium gas are taken over the cycle ABCDA, as shown in the P - T diagram.
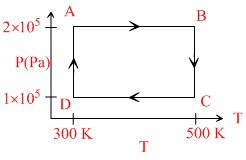
The work done on the gas in taking it from D to A is

The work done on the gas in taking it from D to A is
Options
A.- 414 R
B.+ 414 R
C.- 690 R
D.+ 690 R
Solution
At constant temperature (isothermal process)
WDA = nRT In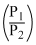 = 2.303 × 2R × 300 log
= 2.303 × 2R × 300 log 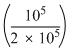
= 2.303 × 600Rlog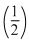
= 0.693 × 600Rlog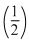 600 R = - 414 R.
600 R = - 414 R.
WDA = nRT In
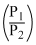 = 2.303 × 2R × 300 log
= 2.303 × 2R × 300 log 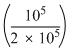
= 2.303 × 600Rlog
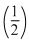
= 0.693 × 600Rlog
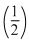 600 R = - 414 R.
600 R = - 414 R.Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Cp - Cv = R correctly applicable to which of the following :-...-If DA and BC are adiabatic curves and AB and CD are isothermal curves then : -If DA and BC are adiabatic curves and AB ...One mole of a monatomic ideal gas is taken along two cyclic processes E → F → G → E and E → F &#...Let , vrms and vp respectively denoted the mean speed, root mean square speed and most probable speed of the molecules i...A diatomic ideal gas is used in a Car engine as the working substance. If during the adiabatic expansion part of the cyc...