ThermodynamicsHard
Question
Let 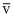 , vrms and vp respectively denoted the mean speed, root mean square speed and most probable speed of the molecules in an ideal monaoatomic gas at absolute temperature T. The mass of molecules is m. Then,
, vrms and vp respectively denoted the mean speed, root mean square speed and most probable speed of the molecules in an ideal monaoatomic gas at absolute temperature T. The mass of molecules is m. Then,
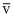 , vrms and vp respectively denoted the mean speed, root mean square speed and most probable speed of the molecules in an ideal monaoatomic gas at absolute temperature T. The mass of molecules is m. Then,
, vrms and vp respectively denoted the mean speed, root mean square speed and most probable speed of the molecules in an ideal monaoatomic gas at absolute temperature T. The mass of molecules is m. Then, Options
A.no molecules can have a speed greater than √2vrms
B.no molecule can have speed less than vp / √2
C.vp < √2 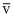 < vrms
< vrms
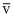 < vrms
< vrmsD.the average kinetic energy of a molecule is 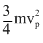
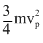
Solution
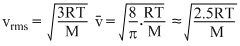
and
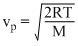
From these expressions we can see that,
vp <
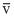 < vrms
< vrms Secondly, vrms =
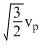 and average kinetic energy of a gas molecule
and average kinetic energy of a gas molecule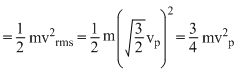
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Air is filled in a tube of the wheel of a car at 27o C and 2 atm pressure if the tube is suddenly bursts, the final temp...On mixing equal volumes of a monoatomic gas and a diatomic gas at the same initial pressure and temprature the ratio of ...5.6 liter of helium gas at STP is adiabatically compressed to 0.7 lit. Taking the initial temperaure to be T1, the work ...In a water fall the water falls from a height of 100 m. If the entire energy of water is converted into heat. The rise i...An ideal gas is taken through a process where dQ = dU - dW. The specific heat of the ideal gas in this process is (dQ = ...