ThermodynamicsHard
Question
A diatomic ideal gas is used in a Car engine as the working substance. If during the adiabatic expansion part of the cycle, volume of the gas increases from V to 32V the efficiency of the engine is
Options
A.0.5
B.0.75
C.0.99
D.0.25
Solution
The efficiency of cycle is
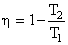
for adiabatic process
TVγ - 1 = constant
For diatomic gas γ =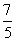
T1V1γ - 1 = T2V2γ - 1
T1 = T2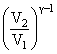
T1 = T2(32)7/5 - 1 = T2 (25)2/5
= T2 × 4
T1 = 4T2.
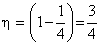 = 0.75
= 0.75
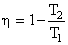
for adiabatic process
TVγ - 1 = constant
For diatomic gas γ =
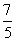
T1V1γ - 1 = T2V2γ - 1
T1 = T2
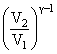
T1 = T2(32)7/5 - 1 = T2 (25)2/5
= T2 × 4
T1 = 4T2.
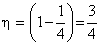 = 0.75
= 0.75Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Fraunh offer line the solar system is an example of :...If ᐃU and ᐃW represented the increases in internal energy and work done by the system respectively in a ther...As shown in the diagram for a closed path ABCA :...A black body is at temperature if 2800 K. The energy of radiation emitted by this body with wavelength between 499 nm an...The volume of a gas expands by 0.25 m3 at a constant pressure of 103 N/m2. The work done is equal to...