Redox and Equivalent ConceptHard
Question
For a first order reaction A → P, the temperature (T) dependent rate constant (k) was found to follow the equation log k = - (2000)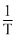 + 60. The pre-exponential factor A and the activation energy Ea, respectively, are
+ 60. The pre-exponential factor A and the activation energy Ea, respectively, are
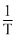 + 60. The pre-exponential factor A and the activation energy Ea, respectively, are
+ 60. The pre-exponential factor A and the activation energy Ea, respectively, areOptions
A.1.0 × 106 s-1 and 9.2 kJmol-1
B.6.0 s-1 and 16.6 kJmol-1
C.1.0 × 106 s-1 and 16.6 kJmol-1
D.1.0 × 106 s-1 and 38.3 kJmol-1
Solution
Given, log K = 6 - 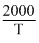
Since, log K = log A -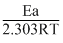 So, A = 106 sec-1 and Ea = 38.3 kJ/mole
So, A = 106 sec-1 and Ea = 38.3 kJ/mole
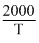
Since, log K = log A -
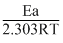 So, A = 106 sec-1 and Ea = 38.3 kJ/mole
So, A = 106 sec-1 and Ea = 38.3 kJ/moleCreate a free account to view solution
View Solution FreeMore Redox and Equivalent Concept Questions
The oxidation state of chromium in the final product formed by the reaction between KI and acidified potassium dichromat...For the redox reaction Zn(s) + Cu2+(0.1 M) → Zn2+(1M) + Cu(s) taking place in a cell, Eocell is 1.10 volt. Ecell f...An aqueous solution of 6.3 g oxalic acid dihydrate is made up to 250 mL. The volume of 0.1 N NaOH required to completely...The oxisation number of carbon in CO2O is :...The oxide of an element contains 67.67% oxygen and the vapor density of its volatilechloride is 79. Equivalent weight of...