Redox and Equivalent ConceptHard
Question
For the redox reaction Zn(s) + Cu2+(0.1 M) → Zn2+(1M) + Cu(s) taking place in a cell, Eocell is 1.10 volt. Ecell for the cell will be 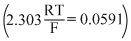
Options
A.1.80 volt
B.1.07 volt
C.0.82 volt
D.2.14 volt
Solution
Ecell = Eocell + 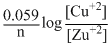
= 1.10 +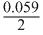 log[0.1] 1.10 - 0.0295 1.07V
log[0.1] 1.10 - 0.0295 1.07V
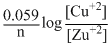
= 1.10 +
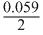 log[0.1] 1.10 - 0.0295 1.07V
log[0.1] 1.10 - 0.0295 1.07VCreate a free account to view solution
View Solution FreeMore Redox and Equivalent Concept Questions
More number of oxidation states are exhibited by the actionoids than by the lanthanoids. The main reason for this is :-...The heat required to raise the temperature of body by 1 K is called...Which of the following will be redox change :-...The oxide of a metal contains 52.91% of the metal. If the formula of the metal oxide is M2O3, then what is the atomic ma...Which of the following is/are peroxide(s)?...