Redox and Equivalent ConceptHard
Question
An aqueous solution of 6.3 g oxalic acid dihydrate is made up to 250 mL. The volume of 0.1 N NaOH required to completely neutralize 10 mL of this solution is :
Options
A.40 mL
B.20 mL
C.10 mL
D.4 mL
Solution
meq. of oxalic acid = meq. of NaOH
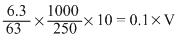
∴ V = 40 mL
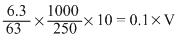
∴ V = 40 mL
Create a free account to view solution
View Solution FreeMore Redox and Equivalent Concept Questions
Which of the following reaction is non-redox?...The following data are obtained in an experiemnt -Given : λm∞(H+) = 350Ω-1 cm2mol-1 ; λm∞(A-)...Preparation of lyphobic sols by chemical method involves...Which of the following will not lose asymmetry on reduction with LiAlH4 :-...In a reaction, calcium phosphate is producing calcium hydrogen phosphate. The equivalent weight of calcium phosphate in ...