Redox and Equivalent ConceptHard
Question
The oxide of an element contains 67.67% oxygen and the vapor density of its volatilechloride is 79. Equivalent weight of the element is :
Options
A.2.46
B.3.82
C.4.36
D.4.96
Solution
Equivalent weight of an element is its weight which reacts with 8gm of oxygen to formoxide.
Thus eq. weight of the given element
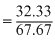 × 8 = 3.82
× 8 = 3.82
Thus eq. weight of the given element
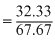 × 8 = 3.82
× 8 = 3.82Create a free account to view solution
View Solution FreeMore Redox and Equivalent Concept Questions
The formula of brown ring complex is [Fe(H2O)5(NO)]SO4. The oxidation state of iron is...The equivalent weight of MnSO4 is half of its molecular weight when it is converted to...Substances which may be oxidized as well as reduced are...When K2Cr2O7 is converted into K2CrO4, the change in oxidation number of Cr is...What is the oxidation state of Xe in Ba2XeO6?...