Question
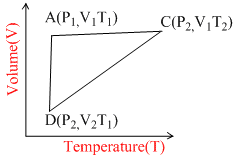
A reversible cyclic process for an ideal gas is shown below. Here, P , V and T are pressure , Volume and temperature , respectively. The thermodynamic parameters q, w, H and U are heat, work, enthalpy and internal energy, respectively
.
The correct option(s) is (are)
Options
qAC = UBC and wAB = P2 (V2 – V1)
Â
wBC = P2 (V2 – V1) and qBC = HAC
HCA < UCA and qAC = UBC
qBC = HAC and HCA > UCA
Â
Solution
AC Isochoric
ABIsothermal
BC Isobaric
# qAC= UBC = nCV (T2 – T1)
WAB = nRT1 ln (v2\v1)Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â A (wrong)
# qBC = HAC = nCP (T2 – T1)
WBC = – P2(V1 – V2)                 B (correct)
# nCP (T1–T2) < nCV(T1 – T2)          C (correct)
HCA < UCA
# D (wrong)
Â
Create a free account to view solution
View Solution Free