Gaseous StateHard
Question
7.5 ml of a gaseous hydrocarbon was exploded with 36 ml of oxygen. The volume of gases on cooling was found to be 28.5 ml, 15 ml of which was absorbed by KOH and the rest was absorbed in a solution of alkaline pyrogallol. If all volumes are measured under same conditions, the formula of hydrocarbon is
Options
A.C3H4
B.C2H4
C.C2H6
D.C3H6
Solution
CxHy + 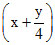 O2 → XCO2 +
O2 → XCO2 + 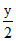 H2O
H2O
7.5 ml 36 ml
36 - 7.5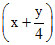 + 7.5 x = 28.5
+ 7.5 x = 28.5
36 - 7.5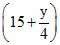 + 7.5 x = 28.5
+ 7.5 x = 28.5
y = 4
x = 2
So formula = C2H4
7.5 ml 36 ml
36 - 7.5
36 - 7.5
y = 4
x = 2
So formula = C2H4
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
500 ml of a hydrocarbon gas burnt in excess of oxygen yields 2500 ml of CO2 and 3 lts of water vapours. All volume being...If two moles of an ideal gas at 546 K occupies a volume of 44.8 litres, the pressure must be -...The ratio between the r.m.s. velocity of H2 at 50 K and that of O2 at 800 K is:...Under which of the following conditions a real gas resembles an ideal gas -...For the diffusion of a gas at pressure P, the rate of diffusion is expressed by:...