Gaseous StateHard
Question
For real gases van der Waals equation is written as
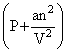 (V - nb) = nRT
(V - nb) = nRT
where ′a′ and ′b′ are van der Waals constants.
I. O2, CO2, H2 and He
II. CH4, O2 and H2
The gases given in set-I in increasing order of ′b′ and gases given in set-II in decreasing order of′a′, are arranged below. Select the correct order from the following:
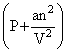 (V - nb) = nRT
(V - nb) = nRTwhere ′a′ and ′b′ are van der Waals constants.
I. O2, CO2, H2 and He
II. CH4, O2 and H2
The gases given in set-I in increasing order of ′b′ and gases given in set-II in decreasing order of′a′, are arranged below. Select the correct order from the following:
Options
A.(I) H2 < O2 < He < CO2 (II) O2 > CH4 > H2
B.(I) He < H2 < CO2 < O2 (II) CH4 > H2 > O2
C.(I) O2 < He < H2 < CO2 (II) H2 > O2 > CH4
D.(I) H2 < He < O2 < CO2 (II) CH4 > O2 > H2
Solution
(I) H2 < He < O2 < CO2 (II) CH4 > O2 > H2
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
At high pressure, the compressibility factor (Z) is equal to :-...The order of ionisation potential between He+ ion and H-atom (both species are in gaseous state) is :-...A sample of H2 collected over H2O at 27oC and a pressure of 591 mm Hg has a volume of 240ml. what volume would the dry H...Which equation show correct from of berthelot eqution....The crystal system of a compound with unit cell dimensions ″a = 0.387, b = 0.387 c = 0.504nm and α = β =...