Chemical Kinetics and Nuclear ChemistryHard
Question
For the following reaction
(CH3)3CCl + H2O → (CH3)3 COH + HCl,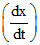 = k[CH3)3CCl], hence, rate determining step can be:
= k[CH3)3CCl], hence, rate determining step can be:
(CH3)3CCl + H2O → (CH3)3 COH + HCl,
Options
A.(CH3)3CCl → (CH3)3C+ + Cl-
B.(CH3)3CCl + H2O → (CH3)3COH + HCl
C.(CH3)3C+ + H2O → (CH3)3COH + H+
D.H+ + Cl- → HCl
Solution
Rate depends only conc. of [(CH3)3CCl] hence rate determening
Step (CH3)3CCl (CH3)3 C+ + Cl-
Step (CH3)3CCl (CH3)3 C+ + Cl-
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
With the help of following reactions, arrange metals A,B,D and E in decreasing order of their reactivity :-(i) B + ANO3 ...A complex reaction 2X + Y → Z, takes place in the following two steps. $X + Y\overset{\quad K_{1}\quad}{\rightarrow}2W$,...In gaseous reactions important for the understanding of the upper atmosphere, H2O and O react bimolecularly to form two ...Order of reactivity is :-...Which of the following graphs represent relation between initial concentration of reactants and half-life for third orde...