Ionic EquilibriumHard
Question
pH of a solution made by mixing 50 mL of 0.2 M NH4Cl and 75 mL of 0.1 M NaOH is :-
[pKb of NH3(aq) = 4.74. log 3 = 0.47]
Options
A.7.02
B.13.0
C.7.02
D.9.73
Solution
NH4Cl + NaOH → NH4OH + NaCl
mmol 50 × 0.2 75 × 0.1
= 10 = 7.5
mmol 10 - 7.5 - 7.5
= 2.5
⇒ This will result in a basic buffer.
⇒ pOH = pKb + log (Salt/Base)
= 4.74 + log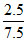 = 4.27
= 4.27
⇒ pH = 14 - 4.27 = 9.73
mmol 50 × 0.2 75 × 0.1
= 10 = 7.5
mmol 10 - 7.5 - 7.5
= 2.5
⇒ This will result in a basic buffer.
⇒ pOH = pKb + log (Salt/Base)
= 4.74 + log
⇒ pH = 14 - 4.27 = 9.73
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The solubility of Pb(OH)2 in water is 6.0 × 10–6 M. The solubility of Pb(OH)2 in a buffer solution of pH = 8 is...The solubility product of AgCl is 1.0 × 10–10. The equilibrium constant of the reactionAgCl(s) + Br–$\rightleftharpoons$...An amount of 0.10 moles of AgCl(s) is added to one litre of water. Next, the crystals of NaBr are added until 75% of the...The composition of an acidic buffer mixture made up of HA and NaA of total molarity 0.29 having pH = 4.4 and Ka = 1.8 &#...What is the pH of solution made by mixing equal volumes of 0.1 N-H2SO4, 0.1 N-HNO3, 0.1 N-HCl?...