Ionic EquilibriumHard
Question
Equal volumes of equimolar solutions of a weak acid and a weak base both having dissociation constant equal to 5 × 10-7 are mixed together. The percentage of hydrolysis of the salt formed by them is :
Options
A.20
B.15.5
C.25.7
D.None
Solution
For hydrolysis of WAWB type salt
h =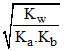 = 0.2
= 0.2
h =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Let the colour of the indicator HIn (colourless) will be visible only when its ionised form (pink) is 25% or more in a s...An amount of 0.15 mole of pyridinium chloride has been added into 500 ml of 0.2 M pyridine solution. Calculate pH of the...The pH value of ordinary water is :...Ka for HCN is 5 × 10-10 at 25oC. For maintaining a constant pH of 9, the volume of 5 M KCN solution required to be ...At infinite dilution the percentage dissociation of both weak acid and weak base is:...