KTGHard
Question
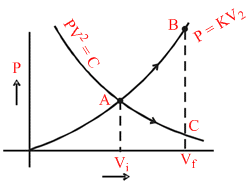
An ideal gas can be expanded from an initial state to a certain volume through two different processes (i) PV2 = constant and (ii) P = KV2 where K is a positive constant. Then
Options
A.Final temperature in (i) will be greater then in (ii)
B.Final temperature in (ii) will be greater then in (i)
C.Total heat given to the gas in (i) case is greater than in (ii)
D.Total heat given to the gas in (ii) case is greater than in (i)
Solution
From equation PV = nRT
PC < PB , VC = VB and TB > TC
WAB > WAC (TB − TA) > TC − TA
So By Ist law
Q = ᐃU + W
Q =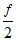 nRᐃT + W QAB > QAC
nRᐃT + W QAB > QAC
PC < PB , VC = VB and TB > TC
WAB > WAC (TB − TA) > TC − TA
So By Ist law

Q = ᐃU + W
Q =
Create a free account to view solution
View Solution FreeMore KTG Questions
When an ideal gas undergoes an adiabatic change causing a temperature change ᐃT(i) there is no heat gained or lost...At N.T.P. the volume of a gas is changed to one fourth volume, at constant temperature then the new pressure will be : (...Find the amount of work done to increase the temperature of one mole of ideal gas by 30oC. if it is expanding under the ...Two containers of same volume are filled with atomic Hydrogen and Helium respectively at 1 and 2 atm pressure. If the te...For an ideal gas, the initial pressure and volume are equal to the final pressure and volume....