KTGHard
Question
Two containers of same volume are filled with atomic Hydrogen and Helium respectively at 1 and 2 atm pressure. If the temperature of both specimen are same then average speed < CH > for hydrogen atoms will be -
Options
A.CH = √2 < CHe >
B.< CH > = < CHe >
C.< CH > = 2 < CHe >
D.< CH > = 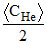
More KTG Questions
For a diatomic gas, change in internal energy for unit change in temperature for costant pressure and volume is U1 and U...250 litre of an ideal gas is heated at constant pressure from 27o C such that its volume becomes 500 liters. The final t...The specific heats of argon at constant pressure and constant volume are 525 J/kg and 315 J/kg respectively. Its density...At 0o C temperature root mean square speed of which of the following gases will be maximum:-...During an experiment, an ideal gas is found to obey a condition = constant [ρ = density of the gas]. The gas is ini...