KTGHard
Question
Find the amount of work done to increase the temperature of one mole of ideal gas by 30oC. if it is expanding under the condition V μ T2/3 (R = 8.31 J/mol - K) :
Options
A.16.62 J
B.166.2 J
C.1662 J
D.1.662 J
Solution
(B)
V = kT2/3
dV =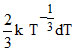
W =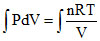 dV
dV
= R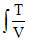 dV = R
dV = R 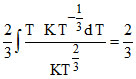 R(T2 − T1)
R(T2 − T1)
=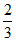 R (30) = 20 (8.31) = 166.2 J
R (30) = 20 (8.31) = 166.2 J
V = kT2/3
dV =
W =
= R
=
Create a free account to view solution
View Solution FreeMore KTG Questions
On increasing the temperature of a gas filled in a closed container by 1oC its pressure increases by 0.4%, initial tempe...The pressure exerted by a gas in P0. If the mass of molecules becomes half and their velocities become double the pressu...The quantity represents (where U = internal energy of gas)...In figure, P-V curve of an ideal gas is given. During the process, the cumulative work done by the gas...An electric fan is switched on in a closed room. The air in the room is :-...