KTGHard
Question
For an ideal gas, the initial pressure and volume are equal to the final pressure and volume.
Options
A.The initial temperature must be equal to the final temperature
B.The initial internal energy must be equal to the final internal energy.
C.The net heat given to an ideal gas in the process must be zero
D.The net work done by an ideal gas in the process may be zero
Solution
T = 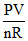 ∴ initial and final temperature are equal
∴ initial and final temperature are equal
as U =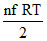 ∴ Uinitial = Ufinal.
∴ Uinitial = Ufinal.
Also the net work done by an ideal gas in the process may be zero.
as U =
Also the net work done by an ideal gas in the process may be zero.
Create a free account to view solution
View Solution FreeMore KTG Questions
At 0o C temperature root mean square speed of which of the following gases will be maximum:-...The speeds of 5 molecules of a gas (in arbitary units) are as follows 2,3,4,5,6 The root mean square speed for these moe...A certain gas is taken to the five states represented by dots in the graph. The plotted lines are isotherms. Order of th...One mole of a gas is subjected to two process AB and BC, one after the other as shown in the figure. BC is represented b...In isothermal process if heat is released from an ideal gas then,...