KTGHard
Question
During an experiment, an ideal gas is found to obey a condition 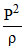 = constant [ρ = density of the gas]. The gas is initially at temperature T, pressure P and density ρ. The gas expands such that density changes to
= constant [ρ = density of the gas]. The gas is initially at temperature T, pressure P and density ρ. The gas expands such that density changes to 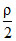
Options
A.The pressure of the gas changes to √2P.
B.The temperature of the gas changes to √2 T.
C.The graph of the above process on the P-T diagram is parabola.
D.The graph of the above process on the P-T diagram is hyperbola.
Solution
Equation of process ⇒ 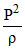 = constant = C .... (1)
= constant = C .... (1)
Equation of State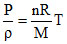 ..........(2)
..........(2)
From 1 and 2 PT = constant ⇒ C is false, D is true.
As ρ-changes to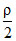 ⇒ P changes to
⇒ P changes to 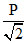 from equation (1) ⇒ A is false.
from equation (1) ⇒ A is false.
Hence T changes to . ⇒ B is true.
Equation of State
From 1 and 2 PT = constant ⇒ C is false, D is true.
As ρ-changes to
Hence T changes to . ⇒ B is true.
Create a free account to view solution
View Solution FreeMore KTG Questions
The pressure exerted by a gas in P0. If the mass of molecules becomes half and their velocities become double the pressu...Let ᐃW1 and ᐃW2 be the work done by the systems 1 and 2 respectively in the previous question then :...Pick the correct statement (s) :...A cyclic process of an ideal monoatomic gas is shown in figure. The correct statement is (are) :...If the pressure of a gas is doubled with constant temperature, then the mean square velocity will become :-...