KTGHard
Question
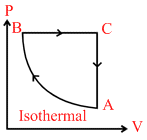
A cyclic process of an ideal monoatomic gas is shown in figure. The correct statement is (are) :

Options
A.Work done by gas in process AB is more than that of the process BC.
B.net heat energy has been supplied to the system.
C.temperature of the gas is maximum in state B.
D.in process CA, heat energy is rejected out by system.
Solution
in cyclic process.
ᐃU = 0 ᐃQ = ᐃW as ᐃW = + ve
∴ ᐃQ = +ve
or Net heat energy has been supplied to the system. in process CA
ᐃW = 0 ᐃU = −ve (As T = decreases
∴ heat energy is rejected out by system
ᐃU = 0 ᐃQ = ᐃW as ᐃW = + ve
∴ ᐃQ = +ve
or Net heat energy has been supplied to the system. in process CA
ᐃW = 0 ᐃU = −ve (As T = decreases
∴ heat energy is rejected out by system
Create a free account to view solution
View Solution FreeMore KTG Questions
The critical temperature of CO2 is -...One mole of an ideal gas at a temperature T1K expands slowly according to the law = constant. Its final temperature is T...A one dimensional gas is a hypothetical gas with molecules that can move along only a single axis. The following table g...For free expansion of a gas in an adiabatic container which of the following is true ?...The molar heat capacity at constant presure of nitrogen gas at STP is nearly 3.5 R. Now when the temperature is increase...