KTGHard
Question
Let ᐃW1 and ᐃW2 be the work done by the systems 1 and 2 respectively in the previous question then :
Options
A.ᐃW1 > ᐃW2
B.ᐃW1 = ᐃW2
C.ᐃW1 < ᐃW2
D.The relation between W1 and W2 cannot be deduced.
Solution
As W.D. by gas in isothermal is more as compare to adiabatic process
∴ ᐃW2 < ᐃW1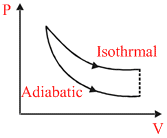
∴ ᐃW2 < ᐃW1

Create a free account to view solution
View Solution FreeMore KTG Questions
In the above question, minimum pressure attainable is...In figure, A and B are two adiabatic curves for two different gases. Then A and B corresponds to :...In a mixture of nitrogen and helium kept at room tempertaure. As compared to a helium molecule nitrogen molecule hits th...The rms velocity of gas molecules is C at temperature T. If the rms velocity is increased to 4 C, then what will be the ...For a gas sample with N0 number of molecules, function N(V) is given by : N(V) = for 0 < V < V0 and N(V) = 0 for V...