KTGHard
Question
Pick the correct statement (s) :
Options
A.The rms translational speed for all ideal-gas molecules at the same temperature is not the same but it depends on the mass.
B.Each particle in a gas has average translational kinetic energy and the equation 1/2 mv2rms = 3/2 kT establishes the relationship between the average translational kinetic energy per particle and temperature of an ideal gas. It can be concluded that single particle has a temperature.
C.Temperature of an ideal gas is doubled from 100oC to 200oC. The average kinetic energy of each particle is also doubled.
D.It is possible for both the pressure and volume of a monoatomic ideal gas to change simultaneously without causing the internal energy of the gas to change.
Solution
Vr.m.s. = 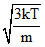
Since PV = nRT therefore P and V both can change simultaneously keeping temperature constant.
Since PV = nRT therefore P and V both can change simultaneously keeping temperature constant.
Create a free account to view solution
View Solution FreeMore KTG Questions
In given figure, let ᐃU1 and ᐃU2 be change in internal energy in process A and B respectively. ᐃQ and ...For an ideal gas, the initial pressure and volume are equal to the final pressure and volume....In a mixture of gases, the average number of degrees of freedom per molecule is 6. The r.m.s. speed of the molecules of ...The average speed of nitrogen molecules in a gas is v. If the temperature is doubled and the N2 molecule dissociate into...According to kinetic theory of gases the relation between internal energy U and absolute temperature T is -...